The complexity of the human brain is unparalleled. Fortunately, thanks to constant progress in neuroscience over the past decades, we have started to make some sense of it. For instance, we now know that neuronal synapses can undergo long-lasting changes in response to their activity and that of nearby neurons, and it is believed this “synaptic plasticity” is one of the key mechanisms behind learning and memory.
Though there are various types of neuronal plasticity, long-term potentiation (LTP) is the most studied one, especially in the hippocampus (brain region associated with learning and memory). Thus far, we know that the hippocampus receives different types of excitatory input from various subcortical regions. However, it is still unclear whether subcortico-hippocampal synapses operating with the glutamate or GABA neurotransmitters undergo any form of LTP and, by extension, if such subcortical inputs contribute to the long-lasting regulation of hippocampal activity.
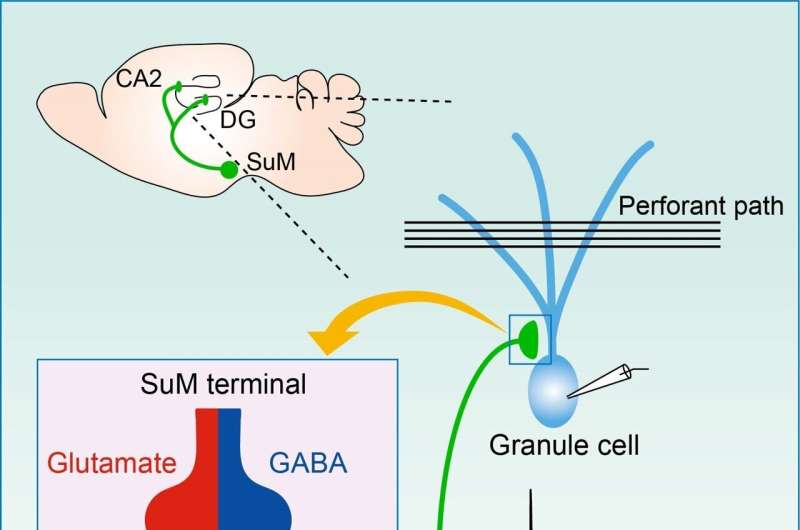
To shed light on this issue, a team of scientists from Doshisha University, Japan, recently conducted a study focusing on the plasticity of the synapses going from the hypothalamic supramammillary nucleus (SuM) to the granule cells (GCs) of the hippocampal dentate gyrus (DG) region.
As explained in their paper published in the journal Cell Reports on December 27, 2022, the team sought to clarify the nature of LTP occurring in SuM–GC synapses, how such LTP is triggered, and what chemicals and proteins are involved. This study was led by Associate Professor Yuki Hashimotodani and included contributions from graduate student Himawari Hirai and Professor Takeshi Sakaba from the Graduate School of Brain Science at Doshisha University.
The team conducted a variety of experiments on the brains of genetically engineered mice whose neurons where modified to fire when exposed to light of specific frequencies (colors). One of the main protocols they used was based on the induction of spike-timing-dependent plasticity by stimulating both pre- and post-synaptic neurons within a critical time window. This allowed them to verify the type of LTP that occurred in the SuM–GC pathways.
The results indicated that the excitatory SuM–GC synapses undergo associative LTP, more specifically a Hebbian type of LTP. Put simply, in this form of plasticity, neurons that fire together strengthen their synaptic connection, making them more likely to fire together again in the future. Moreover, the team found that even though the SuM–GC synapses released GABA and glutamate together, only glutamatergic transmission led to LTP. Further, they identified the synaptic receptors involved in the process.
Overall, their study can help neuroscientists understand the link between the SuM and the DG better. “Our findings suggest that the associative activity of the SuM and the DG can trigger LTP at subcortico-hippocampal excitatory synapses, which can modulate the GC activity and may contribute to SuM-DG-pathway-related learning and memory,” says Dr. Hashimotodani.
In particular, the SuM–DG pathway is interesting since it is pertinent to many brain functions, including spatial memory, sleep/wake cycles, arousal, and locomotion, among others. However, scientists are still in the dark regarding what types of SuM activity patterns lead to LTP in the synapses with GCs.
“It is tempting to speculate that a wide variety of signals from the SuM may participate in LTP induction when associated with DG activity. The behavioral conditions that initiate the correlated activity of the SuM and DG for the induction of LTP will have to be addressed in future studies,” says Hashimotodani.
Doshisha University

